|
2/20/2023 0 Comments Nh3 molecular geometry 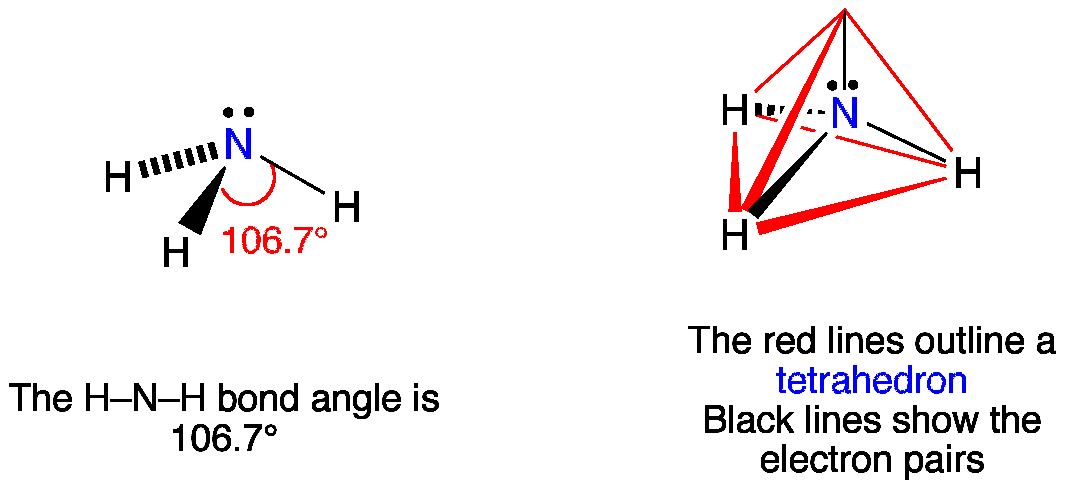
Laboratory 9 Atomic and Molecular Structure 127 Record the bond lengths and bond angles of SFA include units Bond Length Bond Angle F-S F-S-F What is the Electron-Domain Geometry of SF.? Steps 5.1 to 5.4 Record the indicated bond lengths and bond angles of Cyclohexane (Figure a) include units Bond Length Bond Angle C-C-H C-C Laboratory 9 Atomic and Molecular Structure 127 Record the bond lengths and bond angles of SFA.Complete the following table: Substance Lewis Structure Predicted Molecular geometry Bond Angles Polar Bonds Molec. Idealized bond angles for each of the following molecules.ĭetermine the electron geometry, molecular geometry, andĭeviations from the idealized bond angle? Determine the electron geometry, molecular geometry, and.Chrome Extension Get Extension Id With Code Examples.Ef Core Delete All Migrations With Code Examples.How To Log Data To The Flutter Console ? With Code Examples.Find Ip Address Of Virtualbox Vm With Code Examples.The Theory Used To Explain The Behavior Of Solids Liquids And Gases Is With Code Examples.Which Lens Is Converging And Diverging With Code Examples.In Html Onblur And Onfocus Are With Code Examples.Flutter Add Ios Permissions With Code Examples.All the Hydrogen atoms are arranged symmetrically around the Nitrogen atom which forms the base, and the two nonbonding electrons form the tip which makes the molecular geometry of NH3 trigonal pyramidal.2 What type of bond is NH3?Ĭovalent bonds Categories Uncategorized Post navigation NH3 Molecular Geometry Ammonia has a tetrahedral molecular geometry. It is due to the presence of a single non-bonding lone pair of electrons on the nitrogen atom, which acts as a repulsive force on the bonding orbitals. Why is NH3 trigonal planar?Īmmonia (NH3) has a trigonal pyramidal or distorted tetrahedral molecular shape. Ammonia is a compound that has a trigonal pyramidal geometry. 3 hydrogen atoms combine with 1 nitrogen atom to make ammonia. Nitrogen has three electrons in its outermost shell and hydrogen has 1. Trigonal pyramidal shape How is NH3 a single bond?Ī molecule of ammonia (NH3 ) has (a) only single bonds (b) only double bonds (c) only triple bonds (d) two double bonds and one single bond. Hence, three hydrogen atoms each share their 1 electron with nitrogen to form three covalent bonds and make an ammonia molecule (NH3) ammonia molecule. How do you draw the structure of ammonia? How ammonia NH3 is formed?Īmmonia is formed from 3 atoms of Hydrogen and 1 atom of Nitrogen. The nonbonding pair of electrons pushes away from the bonding pairs producing a trigonal pyramidal shape. It is trigonal pyramidal because of the lone pair of electrons associated with the central nitrogen atom. 2), as predicted by the VSEPR theory which valence shell electron pair repulsion. The ammonia molecule has a trigonal pyramidal shape with a bond angle of 107.8° (fig. Structure of ammonia Ammonia by IUPAC name is called azane and it is a compound of nitrogen and hydrogen with the formula NH 3. What is the name of the structure of ammonia? As we’ve seen, a lot of examples were used to address the Ammonia Structure problem. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
